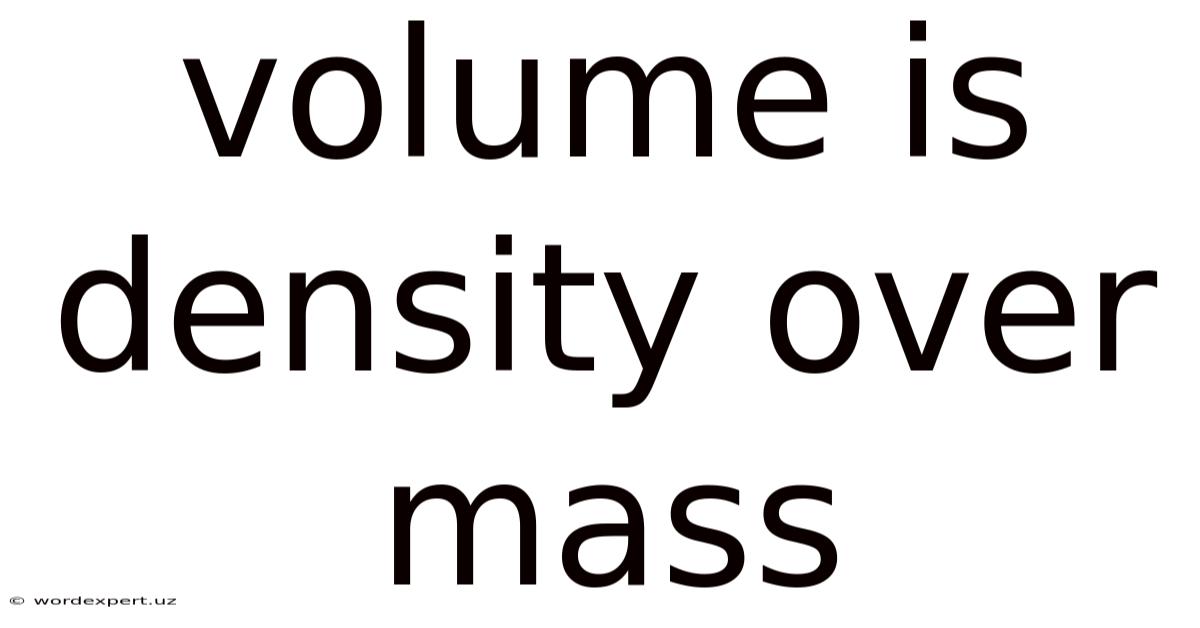The Relationship Between Volume, Density, and Mass: More Than Just a Simple Equation
The statement "volume is density over mass" is incorrect. Practically speaking, the correct relationship between volume, density, and mass is expressed by the equation: Density = Mass/Volume. On top of that, this seemingly small difference highlights a crucial understanding of fundamental physics concepts. This article will dig into the true relationship between these three properties, exploring their definitions, the formula's derivation, applications in various fields, and common misconceptions. We'll also address frequently asked questions to ensure a comprehensive understanding of this vital concept.
Understanding the Fundamentals: Mass, Volume, and Density
Before diving into the equation, let's define each term:
-
Mass: Mass is a measure of the amount of matter in an object. It's a scalar quantity, meaning it only has magnitude and no direction. We typically measure mass in kilograms (kg) or grams (g). Think of it as the "stuff" that makes up an object.
-
Volume: Volume is a measure of the amount of three-dimensional space occupied by an object. It's also a scalar quantity. Common units for volume include cubic meters (m³), cubic centimeters (cm³), and liters (L). Imagine filling a container with water; the amount of water represents the volume of the container That alone is useful..
-
Density: Density is a measure of how much mass is packed into a given volume. It's a scalar quantity representing the mass per unit volume. The formula for density is: Density = Mass/Volume. The standard unit for density is kilograms per cubic meter (kg/m³), but grams per cubic centimeter (g/cm³) is also frequently used. A higher density means more mass is concentrated in a smaller volume. Take this: lead has a much higher density than wood because a given volume of lead has significantly more mass than the same volume of wood Not complicated — just consistent..
Deriving the Density Formula: A Simple Explanation
The formula for density, Density = Mass/Volume, arises directly from its definition. Density tells us how much mass is contained within a unit volume. If we have an object with a mass (M) and a volume (V), dividing the mass by the volume gives us the mass per unit volume – which is the definition of density (ρ) Not complicated — just consistent..
ρ = M/V
This formula is incredibly useful because it allows us to calculate any of the three properties (density, mass, or volume) if we know the other two. We can rearrange the formula to solve for mass or volume:
- Mass (M) = Density (ρ) x Volume (V)
- Volume (V) = Mass (M) / Density (ρ)
Applications of the Density Formula: From Science to Everyday Life
The concept of density and its associated formula have far-reaching applications across numerous fields:
-
Material Science and Engineering: Density is a crucial factor in material selection for various applications. Engineers consider the density of materials when designing structures, choosing materials for aerospace components, or selecting materials for construction projects. Materials with high strength-to-weight ratios (often related to lower density) are preferred in many applications.
-
Chemistry: Density is used extensively in chemistry to identify substances, calculate concentrations of solutions, and determine the purity of samples. The density of a substance is a physical property that can help distinguish between different compounds or mixtures That alone is useful..
-
Geophysics and Oceanography: Density plays a critical role in understanding geological formations and ocean currents. Density variations in the Earth's mantle drive plate tectonics, while density differences in seawater create ocean currents.
-
Medicine: Density measurements are used in medical imaging techniques like bone densitometry (measuring bone density to diagnose osteoporosis) and in assessing body composition.
-
Meteorology: Density differences in air masses are responsible for weather patterns and atmospheric stability. Changes in air density affect the movement of air masses and the formation of clouds and precipitation.
Common Misconceptions and Clarifications
The initial statement, "volume is density over mass," is a common misunderstanding. Plus, it’s crucial to remember the correct relationship: Density = Mass/Volume. This misunderstanding often stems from incorrectly rearranging the formula.
Another misconception is assuming that density is always constant for a given substance. That's why while density is often treated as a constant under standard conditions (temperature and pressure), it can vary depending on these conditions. To give you an idea, the density of water changes slightly with temperature. Gases are particularly susceptible to density changes with changes in temperature and pressure Turns out it matters..
Finally, it’s important to understand the difference between mass and weight. Mass is a measure of the amount of matter, while weight is a measure of the force of gravity acting on that mass. Density is related to mass, not weight That's the whole idea..
Working with Units: Consistency is Key
When using the density formula, it's crucial to ensure consistent units. Plus, if you use kilograms for mass and cubic meters for volume, the density will be in kilograms per cubic meter. Using a mix of units (e.Because of that, g. On the flip side, , grams for mass and cubic meters for volume) will lead to an incorrect result. Always convert all measurements to a consistent system of units (like the SI system) before performing any calculations.
Advanced Concepts: Density and Buoyancy
The concept of density is intimately linked to buoyancy. This principle explains why ships made of steel (a dense material) can float on water. Because of that, if the density of the object is less than the density of the fluid, the buoyant force is greater than the object's weight, and the object floats. In practice, archimedes' principle states that an object immersed in a fluid experiences an upward buoyant force equal to the weight of the fluid displaced by the object. If the density of the object is greater than the density of the fluid, the object sinks. The overall density of the ship, including the air inside, is less than the density of water.
Frequently Asked Questions (FAQ)
Q: What happens to the density of an object if its mass increases while its volume remains constant?
A: The density will increase. Since density is directly proportional to mass (at constant volume), an increase in mass results in an increase in density Turns out it matters..
Q: What happens to the density of an object if its volume increases while its mass remains constant?
A: The density will decrease. Since density is inversely proportional to volume (at constant mass), an increase in volume results in a decrease in density.
Q: Can density be negative?
A: No, density cannot be negative. Mass and volume are always positive quantities, and their ratio (density) must therefore also be positive Simple as that..
Q: How can I measure the density of an irregularly shaped object?
A: You can measure the mass of the object using a balance. To measure the volume, you can use the water displacement method. Submerge the object in a graduated cylinder filled with a known volume of water. The increase in water level represents the volume of the object But it adds up..
Conclusion: Mastering the Density Formula
Understanding the relationship between mass, volume, and density is fundamental to many scientific and engineering disciplines. By grasping the underlying concepts and avoiding common misconceptions, you can confidently apply this formula to a wide range of applications. Remember the importance of consistent units and the interconnectedness of density with other physical phenomena like buoyancy. Worth adding: the formula Density = Mass/Volume is a powerful tool for solving problems involving these properties. This comprehensive understanding will equip you to tackle complex problems and delve further into the fascinating world of physics and material science Simple as that..
Most guides skip this. Don't.